Post technical meeting statement on FH paediatric screening
FH paediatric screening in the EU and the wider Europe is achievable, with the right political will, smart investment and genuine partnership that include patients and citizens as equal players.
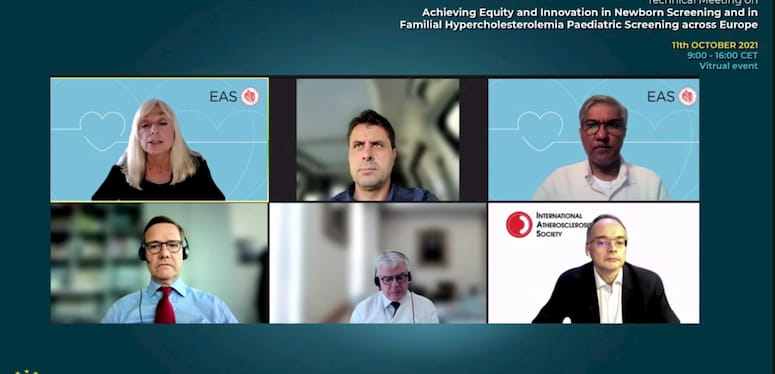
Towards a consensus on FH screening in Europe, A dialogue with key stakeholders Moderator: − Prof. Lale Tokgözoğlu, Past President, European Atherosclerosis Society Panel: Dr. Iñaki Gutiérrez-Ibarluzea, Member of Technical Advisory Group, World Health Organization, Past President, HTAi; Prof. Kausik Ray, President, European Atherosclerosis Society; Prof. Martin Halle, President, European Association of Preventive Cardiology (ESC); Prof. Fausto Pinto, President World Heart Federation; Prof. Raul Santos, President, International Atherosclerosis Society
The overriding message from the high-level technical meeting on FH paediatric screening today was also “The time to act is now” – The topic has been on the agenda since a WHO meeting in Geneva in 1998 with, arguably, very patchy progress since then – Today, there was a very strong consensus on and affirmation of commitment to ensure FH Paediatric universal, cascade and opportunistic screening to all who need and choose it, across Europe.
The scientific name Familial Hypercholesteroleamia – in short FH – describes a genetic variation in one out of 250 to 300 Europeans that increases levels of harmful cholesterol (LDLc) in their blood to life-threatening levels. When untreated, the likelihood of attracting cardiovascular disease at an early age is high, with premature heart attacks and death a frequent outcome.
Speakers and contributors shared a powerful journey during the course of this afternoon – starting with the stark reality of patients living with FH and living with tragedies that could have been avoided: understanding the unmet need out there for hundreds of thousands of patients and citizens with FH – less that 10% of Europeans with FH are currently diagnosed – and less than 5% of Europe’s children. The meeting discussed the wider cardiovascular disease community at European level, how this is uniting, and leading, to drive long awaited change, and how the FH community can be part of this.
Participants heard a global perspective, and how the Global Call for Action has helped to shape the European agenda – and learned from the Global Registry, the powerful evidence base for FH paediatric screening. The meeting explored good practices in FH paediatric screening in several EU countries: Slovenia; the Netherlands; the Czech Republic and France, which can inspire the community and policy makers moving forward, showing what is really possible and its impact on people’s lives. On the basis of these good practices, a draft series of key recommendations have been shared which map out next steps and which will be refined, following consultation at the meeting and beyond. In the session on consensus building, close FH allies and fellow stakeholders discussed these and were unequivocal in their view that we can build on the achievements to date, we do not need to reinvent the wheel, but we do need to address fragmentation and piecemeal approaches. FH paediatric screening in the EU and the wider Europe is achievable, with the right political will, smart investment and genuine partnership that include patients and citizens as equal players. And this was reiterated in the final session of the day, in which policy makers discussed the role of national governments, the role of the EU institutions and the role of groups like FH Europe.
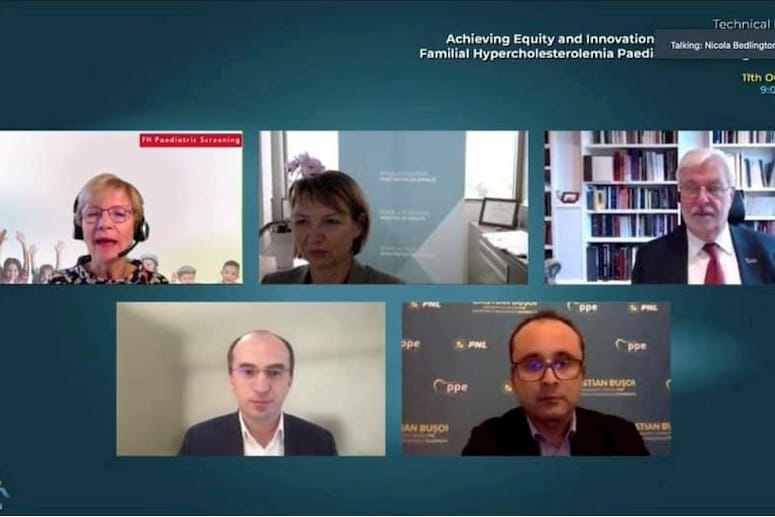
High level political debate moderated by Ms. Nicola Bedlington, FH Europe Sr. Advisor (top left) with Dr Mojca Gobec, Head of the Sector for Disease and Injury Prevention at the Public Health Directorate, Slovenian Ministry of Health, Slovenian Presidency of the EU Council, John Reeve, Chair of FH Europe, Dr Marius Geanta, Trustee, FH Europe and President, Centre for Innovation in Medicine (bottom left), Cristian-Silviu Buşoi, MEP and Chair, Industry, Research and Energy (ITRE) Committee
Véronique Schaal, an FH patient representative from France stated at the meeting “My feeling today is that early testing is crucial; awareness and understanding is crucial – and addressing mental health impact of fear, isolation and taboo is crucial. If we can protect our children and future generations from this, through screening, the impact will be enormous”

Véronique Schaal, an FH patient representative from France
Professor Urh Groselj, Scientific Advisor to FH Europe said:
“Familial hypercholesterolemia is the most common life-threatening inherited metabolic disorder in humans, which is on one hand extremely under-diagnosed, but on the other easily treatable once diagnosed. Early prevention of cardiovascular complications in people with familial hypercholesterolemia needs to start in childhood if we want to normalize their risk for complications in the long run. Sadly, only few percent of all the children with familial hypercholesterolemia are diagnosed in Europe and globally, and thus at high risk for cardiovascular complications later-on. We also know an effective answer to that: to screen familial hypercholesterolemia in all children. We need to form a coalition of all the stakeholders with the aim to make childhood familial hypercholesterolemia screening reality. I am very pleased that the Slovenian technical meeting has helped start this “ball rolling”, not just in Europe but globally. Urgent action is needed.”
Magdalena Daccord, CEO, FH Europe underlined “A common vision and a common mission has been agreed which means within three years we can be well on the way to a systemic approach to FH paediatric screening in all European countries – we will have a robust action plan – and we all have a distinct and collective role to play in this. We are very pleased that the Slovenian EU Council Presidency has championed this, to mobilise member states, and we look to the upcoming French Presidency to pick up the baton. The message from the Commission is loud and clear -there are opportunities, through new funding programmes to nurture and support EU cooperation on FH screening – and it is also clear we have the backing of the vibrant European Parliament Heart Group.”
John Reeve, President of FH Europe said “As President of FH Europe, I commit my organisation to being a convener, an accelerator and a watchdog, to ensure that we deliver on our commitments today. to make FH paediatric screening a reality in Europe”.
For further information, please contact Magdalena Daccord, CEO, FH Europe
Link to the programme: https://www.nbs-fh-screening-si2021.eu/
Link to the backgrounder: https://fheurope.org/latest-news/the-time-is-now-cardiovascular-disease-as-an-eu-priority/
A recording of the meeting is available at https://www.youtube.com/playlist?list=PL4l_jdUr544pH135ORJ8YhFIFtdv5Atre
Information about FH Europe
FH Europe actively works to secure early identification and diagnosis of FH (Familial hypercholesterolaemia) patients to prevent early cardiovascular disease. FH Europe aims at sharing information and best practice across Europe, work with experts to focus on themes and topics of interest to the patients and families we represent and supporting development of newer or smaller patient groups and individuals wanting to start a patient group.
FH Europe is supported by an educational grant from Amgen Limited, Sanofi, Regeneron, Akcea Therapeutics Inc. and Amryt
Site by: Vovi Web Design

